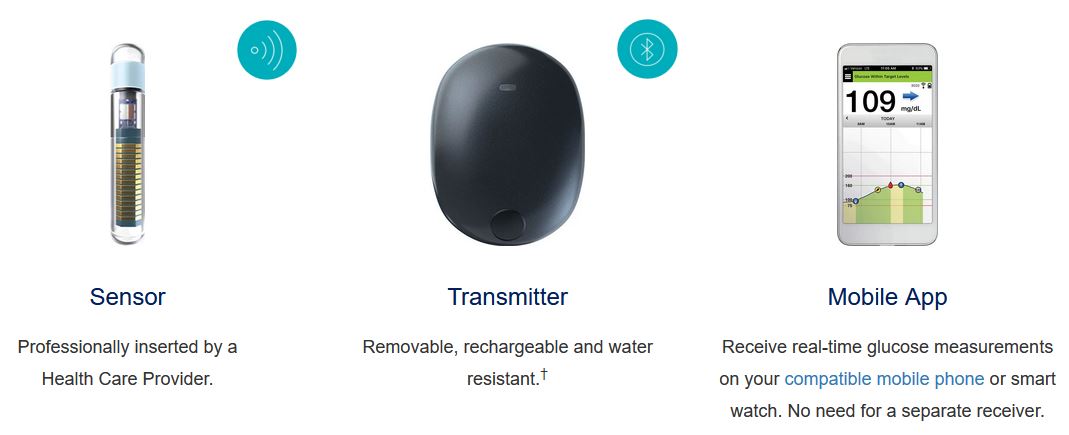
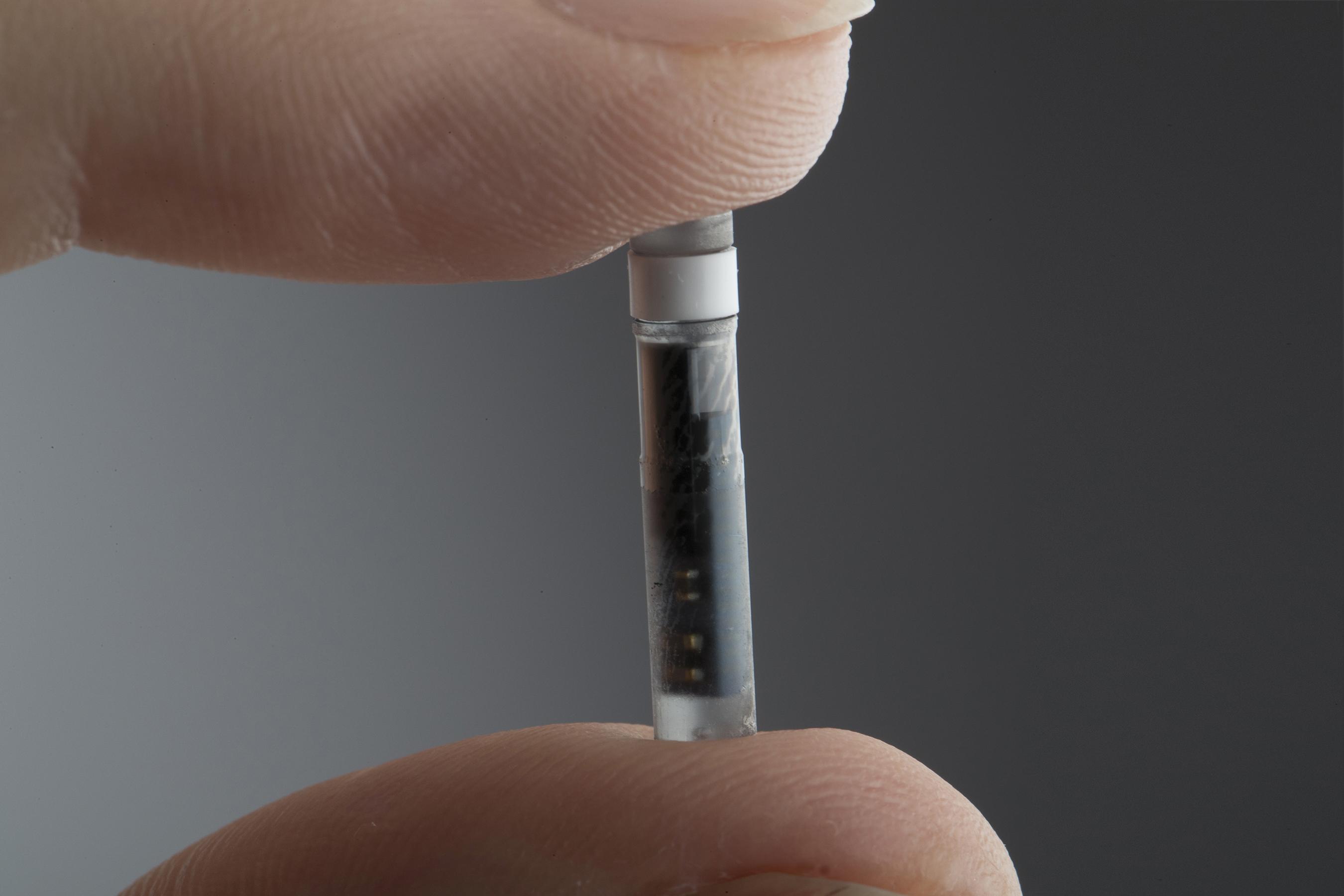
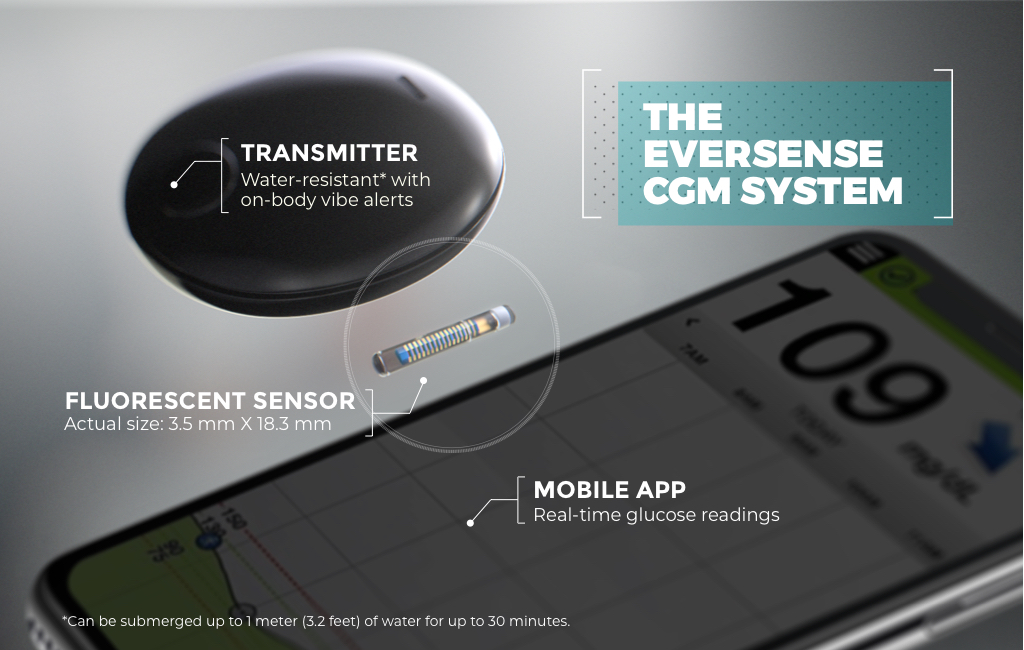
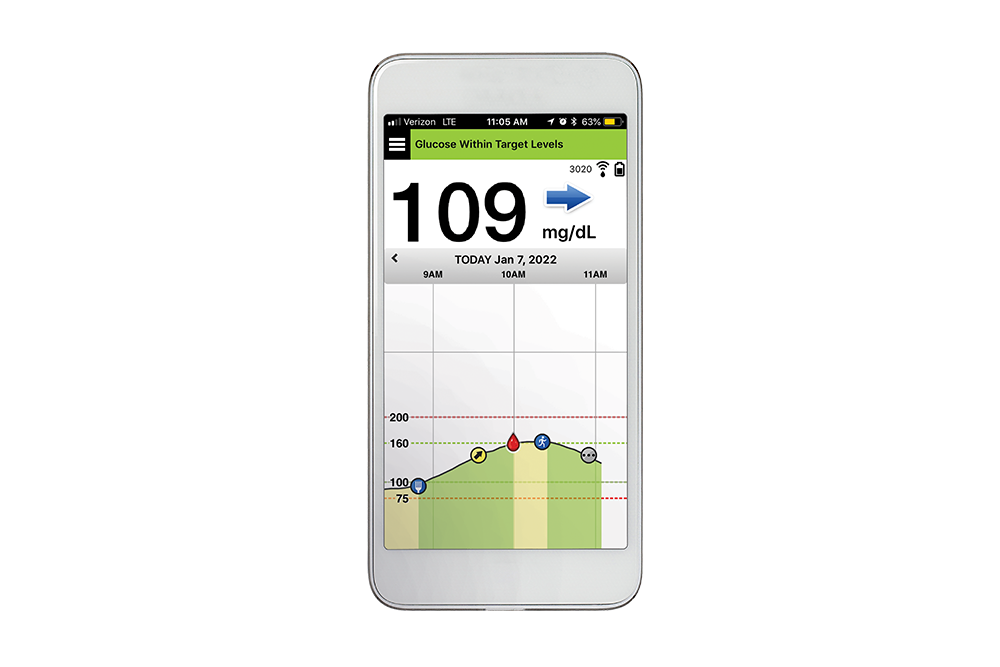
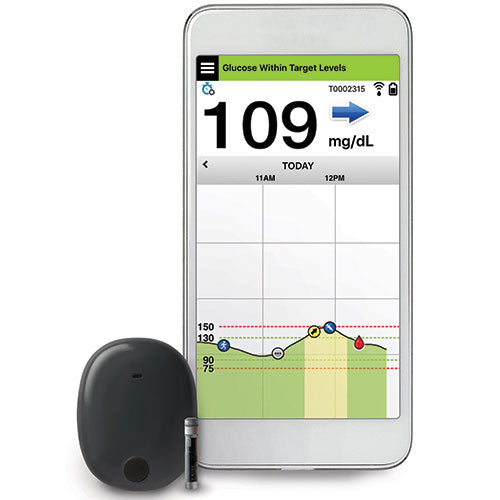
Implantable glucose sensor featuring IDT sensing technology awarded CE Mark - Medical Design and Outsourcing

Eversense sensor placement. (a) The positioning guide for the Eversense... | Download Scientific Diagram

Eversense sensor. CM, centimeter; DXA, dexamethasone acetate; PMMA,... | Download Scientific Diagram